OEM & Manufacturing
17 min read
February 04, 2026
Home > Blog > OEM & Manufacturing > The Science of Fresh Breath Sprays: Volatility, Flavor Binding, Shelf Life

Source: Pexels
Breath sprays look simple. You just press, spray, and get instant freshness. Behind that simplicity lies formulation complexity that determines whether your product delivers consistent results after six months on the shelf or becomes a complaint magnet as volatile compounds evaporate prematurely.
Fresh breath spray formulation involves managing chemistry that actively fights containment. Menthol wants to escape. Essential oils oxidize under light exposure. Alcohol evaporates through valve seals. Flavor stability drops 20–40% over the typical shelf life unless flavor binding systems hold volatile compounds in suspension. These aren’t lab curiosities—they’re margin threats that show up as customer returns, retailer rejections, and inventory write-offs.
Breath spray stability challenges exceed those of bottled mouthwash or sealed sachets because spray formats maximize exposure to air. They also create headspace dynamics that accelerate evaporation and rely on mechanical valves, introducing leakage variables. A single sachet packet of mouthwash seals its contents until use. A spray bottle loses volatile compounds every time someone presses the actuator, and every day the product sits in inventory.
This guide explains how flavor binding systems, packaging choices, and stability testing protocols affect commercial outcomes: unit economics, MOQ thresholds, oral spray shelf life, and distribution feasibility. Whether you’re evaluating oral spray formulation for hotel amenity kits, gym lockers, or a retail launch, understanding volatility control determines whether your spray product performs profitably or creates operational problems.
In This Article
Volatile loss weakens fresh breath sprays after 8 months in inventory, causing inconsistent results and fewer repeat buys. Here’s how it happens:
Alcohol evaporation in sprays occurs in two ways: through tiny valve leaks and air space inside the bottle. Each spray lets air in, so alcohol and scent compounds evaporate. Between uses, bottle pressure pushes volatile bits out through valve gaps.
Sprays with alcohol (usually 15–30% ethanol) lose it faster than alcohol-free ones. Ethanol is super volatile and drags menthol, eucalyptol, and oils with it. Losing just 5% alcohol over six months wipes out 15–25% of cooling and aroma power because these scents stick to the alcohol.
This hits freshness hard. Buyers at month one feel a strong minty chill. By month seven, it’s weak and dull, even if not expired.
Breath spray stability hinges on headspace—the empty space above the liquid to the cap. Big gaps speed up evaporation by giving vapors room to grow. Tiny gaps cut evaporation but build pressure in heat, pushing liquid past seals.
Bottle material choice is key. Plastic (PET, HDPE) lets some vapor slip through. Glass or aluminum blocks it better. PET works for travel and low costs, but volatiles escape walls over time. Glass holds flavors longer but adds weight, shatter risk, and shipping expenses.
Valve seals differ by maker. Top ones have layered gaskets and precise parts to stop leaks. Cheap ones save $0.05–$0.10 each but cause sticky outsides, fewer sprays, and quick flavor fade.
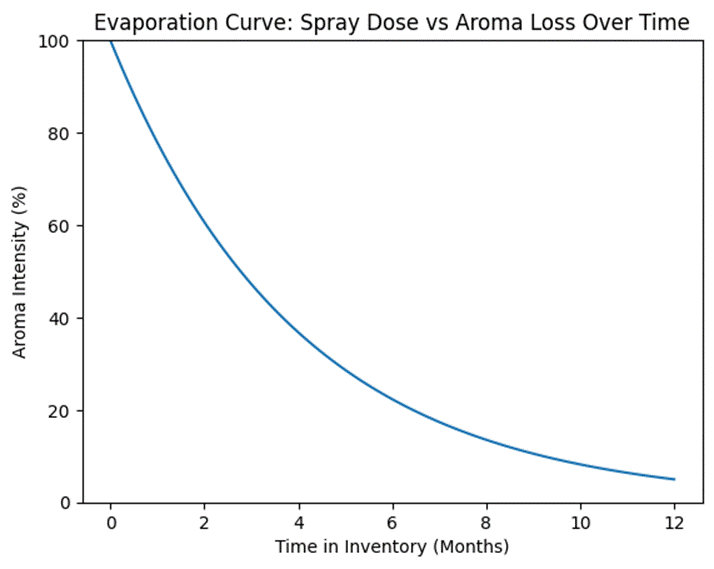
Evaporation Curve Diagram (spray dose vs aroma loss over time)
Flavor binding systems slow volatile loss by trapping aroma compounds in molecular structures that release gradually rather than evaporating immediately.
Oral spray formulation uses three main approaches to control volatility:
These choices boost stability and change flavor burst speed, lasting power, and mouth sensation (light vs. oily coating).
Fresh breath spray formulations must balance instant impact with lasting freshness. Fast-release systems give strong cooling in seconds but fade quickly. Slow-release formulas take 10–15 seconds to peak and keep freshness 20–30 minutes versus 5–10. This timing influences buyers differently: retail shoppers testing in-store want immediate intensity, DTC subscribers value longer-lasting effect, and hotel amenity users need quick freshness without a lingering strong menthol taste.
| Binding System Type | Volatility Control | Sensory Impact | Cost Per Unit | Typical OEM MOQ Impact |
| No binding (free oils) | Low | Immediate, fades fast | Lowest | Standard MOQ |
| Solubilizer networks | Moderate | Quick onset, moderate duration | +$0.05–0.15 | Standard to moderate |
| Polymer complexes | High | Gradual onset, sustained effect | +$0.15–0.35 | Higher MOQ |
| Carrier matrices | Moderate-high | Balanced onset/duration | +$0.08–0.20 | Standard MOQ |
Stability investments affect unit economics in non-obvious ways. Those ways include:
A basic breath spray formula using standard solubilizers costs $0.40 per unit but loses 35% of its flavor over 12 months. With an average shelf turnover of seven months and 15% of inventory sitting longer, customers get inconsistent experiences.
Upgrading the formula with polymer binding adds $0.15 per unit but cuts flavor loss to 15% over 12 months. This, in turn, results in fewer returns, higher ratings, and stronger repeat purchases.
Complex binding systems require more mixing stages, longer hold times, and sometimes specialized equipment. Fresh breath spray formulation using cyclodextrin complexes might require 10,000–15,000 units minimum compared to 5,000–8,000 for standard formulas. This MOQ difference matters for brands testing new flavors or entering markets with uncertain volume. Download the Formula Brief Template to define binding system preferences, shelf-life targets, and MOQ constraints before requesting OEM quotations.

Source: Pexels
Oral spray shelf life expectations must account for format-specific degradation pathways.
Breath spray shelf-life testing follows standard accelerated protocols but adds actuation testing. Results often show unopened bottles maintain 90% flavor intensity at 12 months, while bottles actuated 50 times drop to 75%.
Alcohol-free formulas typically achieve a 12–18-month shelf life with proper binding systems. Alcohol-based formulas can reach 18–24 months but require tight valve seals and low-permeation packaging.
Breath spray stability degrades faster under light exposure. Clear bottles in retail display environments experience 20–30% faster degradation than amber or opaque containers. Temperature cycling during shipping stresses valve seals and accelerates volatile escape. Products shipped internationally face extremes beyond standard laboratory testing.
Format selection determines whether breath spray programs succeed commercially.
Single sachet packet mouthwash seals contents in multi-layer foil until use. Spray bottles expose contents to air repeatedly and rely on valve integrity.
For hotel amenity programs, sachets avoid housekeeping complaints about leaking bottles. For gym locker distribution, sprays win because members want quick application without finding a sink.
Match packaging to where it’s sold: hotels want leak-proof, airlines need TSA-approved sizes, gyms require tough, moisture-resistant bottles.
Single Sachets/Foil Packs – Ideal for airlines, hotels, conference kits.
Leak-proof, lightweight, TSA-friendly (as wipes). Cost: $0.15–$0.35 each. Downsides: One-time use, trash issue, feels cheap.
Small Spray Bottles (10–20ml) – Great for gyms, travel, retail samples.
Portable, reusable (30–50 uses), TSA-ok. Cost: $0.70–$1.50 each. Issues: Leaky valves, flavor fades, pricey per ml.
Standard Bottles (30–50ml) – Suited for hotel shops, wellness programs, retail.
Lasts 2–4 weeks, good value. Downsides: Too big for TSA, evaporates if not full.
Bulk Dispensers – Perfect for gyms, offices, spas.
Cheap per use ($0.02–$0.05), less waste. Drawbacks: Germs spread easily, needs refills and cleaning—hygiene worries post-COVID.
| Channel | Recommended Format | Why It Works | What Fails |
| Airlines | Sachets | TSA-friendly, zero leakage | Bottles (pressure issues) |
| Hotels | Sachets or 30ml bottles | Housekeeping-safe, perceived value | Bulk dispensers (maintenance) |
| Gyms | 20ml bottles or wall dispensers | Durability, quick access | Sachets (disposal logistics) |
Budget valves deliver inconsistent spray patterns. Premium valves maintain consistent atomization, ensuring each spray delivers a similar sensory impact. Here, consistency matters for products making quantitative claims like “200 sprays per bottle.”
Source: Pexels
Breath spray quality control faces distinct contamination pathways.
Breath spray microbial contamination risks increase when users often touch the spray nozzle with their lips or tongue. Alcohol-based sprays naturally keep water activity low, which helps prevent microbial growth. Alcohol-free sprays, however, rely on added preservatives to stay safe, making formulation and testing more critical.
Preservative effectiveness must be verified under real-world conditions, testing not only unopened bottles but also sprays after repeated use. This ensures the product remains safe and free from harmful bacteria throughout its shelf life, even after multiple actuations. Proper preservative selection and validation are essential for user safety, regulatory compliance, and maintaining product quality.
Oral care QA documentation should include stability summaries, microbial reports, batch records, and packaging compatibility notes. Spray-specific testing should also document actuation consistency. Request Flavor Sample Kit to compare binding system performance across different storage conditions and use patterns before committing to formulation.
Lab formulas pass tests but flop at scale (e.g., 50,000 units). Labs control conditions; factories face heat swings in filling, storage, and shipping (up to 140°F).
OEMs with strong systems share this easily. Hesitation means risks ahead.
Fresh breath spray formulation benefits greatly from OEM collaboration. Engineers help buyers assess stability risks based on shelf-life goals, distribution regions, and usage patterns. They recommend binding systems that fit MOQ requirements, adjust release profiles, and test packaging compatibility before production.
To review stability planning and MOQ models, book a Formulation Call. This process also covers packaging compatibility and ensures your brand is documentation-ready before finalizing specifications.

Source: Pexels
Hotels prioritize leakage prevention and conservative shelf-life claims. Alcohol-free formulations simplify international compliance.
Gyms want immediate freshness and a safety perception. Alcohol-free positioning aligns with wellness branding and cost control.
Event organizers prioritize immediate sensory impact and transport stability over long-term shelf life.
Breath spray regulatory claims determine how a product is classified and overseen by authorities. Cosmetic claims emphasize temporary benefits such as freshening breath, adding fragrance, or enhancing sensory experience. These avoid FDA drug pathways, premarket approvals, and most rigorous testing.
Therapeutic claims, even casually worded, suggest disease treatment or health effects. Statements about treating halitosis, killing bacteria, or preventing oral disease can trigger OTC drug classification, requiring stricter compliance, testing, and labeling.
Breath spray labeling rules highlight words and ingredient descriptions that increase regulatory risk. Terms like antimicrobial, antibacterial, or disease-prevention, especially when paired with essential oils or actives linked to oral health, signal therapeutic intent.
Describing the same ingredients in cosmetic or sensory terms, such as “refreshing” or “soothing,” preserves cosmetic classification, simplifies retailer approval, eases international distribution, and streamlines documentation review. Careful wording protects listing acceptance, maintains compliance, and supports long-term brand growth.
Schedule a breath spray formulation consultation to review stability planning, binding system options, packaging format selection, MOQ implications, and compliance requirements before committing to production.
Schedule a consultation to discuss your industry needs, volume requirements, and custom formulation options.
Trusted by 200+ brands across hospitality, retail, and travel sectors